May 9, 2026
What Is Liposomal Vitamin C and Why Do I Need It?
Ascorbic acid is enclosed in phospholipid bilayers obtained from non-GMO lecithin sources in liposomal vitamin C powder, which is a biotechnological improvement in nutrient delivery. Because this encapsulation looks like the cell membranes in the human body, vitamin C can get through stomach problems and be absorbed up to 8 times better than in powder form. The technology solves important bioavailability problems that make regular vitamin C formulations less effective. This makes it necessary for companies making high-performance immune support, antioxidant, and health goods.
Understanding Liposomal Vitamin C Powder
The main difference between liposomal versions and regular vitamin C is how the vitamin is delivered. Standard ascorbic acid is absorbed by the body through active transport mechanisms in the small intestine. These become saturated at amounts above 200 mg, so bigger doses are only absorbed by about 20% of the body. The phospholipid bilayers create vesicles that fuse with intestinal cell membranes via endocytosis or direct membrane fusion.
The Science Behind Enhanced Absorption
Nano-encapsulation technology is used by our EncapsWell™ platform. This technology surrounds vitamin C molecules with phospholipid layers that are 50–200 nanometers thick. These structures keep ascorbic acid from being broken down by stomach acid and digestive enzymes. This keeps the nutrients intact until they are taken up by cells. Clinical tests have shown that this method produces significantly higher plasma concentrations than non-encapsulated forms, resulting in sustained blood levels.
Why Bioavailability Matters for Product Formulation?
When you make functional drinks or vitamins, the active ingredient has to get to the right cells to do what it's supposed to do. Poor bioavailability directly compromises product efficacy, leading to consumer dissatisfaction and brand erosion. Liposomal packaging makes sure that ≥99% of the vitamin C content stays effective. This closes the gap between what the label says and what happens in the body. This is especially helpful for immune-modulating formulas, collagen-synthesis support products, and antioxidant mixes that need to make sure they send their ingredients consistently to cells in order to stand out in the market.
Stability Advantages for Supply Chain Management
In addition to being easily absorbed, liposomal vitamin C powder has better stability profiles that are needed for world spread. When stored properly, our recipes keep more than 95% of their effectiveness for 24 months. They don't break down like most powders do when they get wet or oxidize. This steadiness makes the food last longer, cuts down on waste, and makes it easier for brands to manage their supplies in multiple regions. Because it dissolves in water, it can be easily added to beverage systems, fizzy tablets, and ready-to-mix sachets without causing precipitation or sticking.
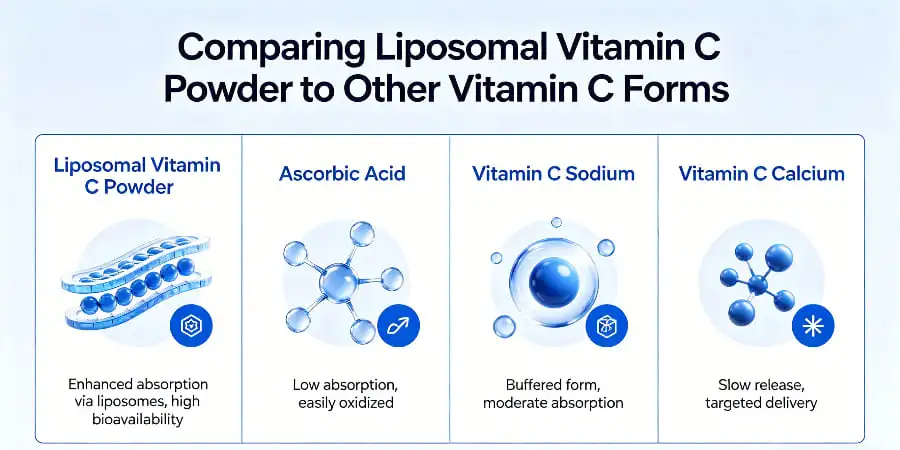
Comparing Liposomal Vitamin C Powder to Other Vitamin C Forms
Before making a purchase choice, it's important to know how different forms of vitamin C work in key areas. Ascorbic acid comes in solid form in traditional pills, capsules, and powders that are not enclosed. This makes it difficult for them to dissolve in stomach acid. Liquid liposomal goods have similar solubility benefits, but they are harder to store, cost more to ship because they are heavier, and need preservatives that go against the clean-label positioning.
Absorption Rates and Gastrointestinal Tolerance
Due to high acidity and osmotic effects in the intestine lumen, standard vitamin C powders in amounts above 1,000 mg often make people feel sick, have stomach pain, or have loose stools. Liposome encapsulation mitigates these irritants by protecting the vitamin until it reaches the intestinal membrane for fusion-triggered release. Controlled absorption tests show that liposomal versions get vitamin C levels in the blood that are similar to intravenous administration at the same amounts, but without the stomach problems that make it hard for people to take regular oral forms.
Chewable pills and sweets are convenient for customers, but they limit the number of doses they can take and often need extra sugars or sweeteners, which goes against what the brand says about being healthy. Our powder form lets you customize the dose, ranging from 500 mg to 2,000 mg per serving. This lets you market a wide range of products, from daily maintenance formulas to therapeutic-level immune boosts. As more and more global markets demand vegan, non-GMO, and allergen-free products, the fact that these don't have any of the fillers needed for pill compression or gummy grids meets those needs.
Cost-Effectiveness Analysis for B2B Procurement
Liposomal vitamin C powder costs more per kilogram than regular ascorbic acid, but because of the absorption multiplier effect, lower amounts can still achieve the same or better health results. A liposomal dose of 500 mg gives cells the same amount of vitamin C as 4,000 mg of regular powder, which changes cost-per-efficacy figures in a big way. When you buy in bulk from OEM/ODM partners, you can get even better returns. This is especially true when you combine their products into full product lines that include pills, tablets, and liquids from a single source.
How to Choose the Best Liposomal Vitamin C Powder for Your Business?
Making choices about where to get things depends on checking technical specs, making sure suppliers follow the rules, and making sure suppliers can support production that can be scaled up. Important factors start with capsule efficiency measures. Liposomes that aren't made well can break during processing or storage, which takes away any bioavailability benefits. Manufacturers you can trust give you information on particle size distribution, zeta potential readings, and encapsulation rates that have been checked by a third party.
Certification and Quality Assurance Priorities
Compliance with regulations can open or close market entry. Our Wellgreen factory keeps up with cGMP, ISO 9001/22000, HACCP, FSSC22000, and HALAL/Kosher certifications. This makes sure that our goods meet the standards of more than 50 countries, such as FDA rules for US markets, EU Novel Food rules, and ANVISA rules for sales in Brazil. Each batch goes through strict stability tests that can be tracked from where the raw materials come from to where they are packaged at the end. This gives us the Certificates of Analysis and stability reports we need for regulatory applications and customer checks.
Vegan and non-GMO labels reach groups of customers that are growing quickly. When lecithin comes from sunflowers instead of soy, allergen worries go away and plant-based recipe requirements are met. Third-party verification through groups like the Non-GMO Project adds a legitimate seal of approval that helps a brand place itself in higher-end markets.
Supplier Reliability and Customization Flexibility
How long a partnership lasts relies on more than just the strength of the ingredients. Our 100-metric-ton annual capacity works for both new brands with low minimum order numbers and well-known companies that need a steady supply of large goods. OEM/ODM services include custom mixes that combine vitamin C with active ingredients that work well together, such as zinc, quercetin, elderberry, or collagen peptides. This lets you make unique SKUs without having to deal with multiple vendors.
Technical support is equally critical. Our PhD-led research and development team in San Diego helps with formulation, making sure the product is stable in hot temperatures, and bioavailability modeling, which speeds up the product development process. Localized US storage through EmerWell makes sure that samples are delivered quickly and that contact is responsive during North American business hours. This takes care of some of the logistical problems that come up when buying things from other countries.
Practical Guidance on Using Liposomal Vitamin C Powder
For inclusion into finished goods to work well, dosing rules, preparation methods, and safety factors must be followed to get the best results while protecting consumers. For immune support, 500 mg to 1,000 mg daily is usually enough. For antioxidant or collagen-synthesis purposes, 1,500 mg to 2,000 mg may be needed, but it should be split up into several doses.
Formulation Best Practices
Liposomal vitamin C powder works best in pH levels between 5.5 and 7.0, which are normal to slightly acidic. For beverage uses, gentle mixing is better than high-shear blending because it keeps the structure of the liposomes. When effervescent delivery systems dissolve, they make a small amount of acidity that improves taste while keeping the structure of the capsules. For tablet or capsule formats, the powder can be processed using normal encapsulation or direct compression equipment without any special changes. During production, moisture-controlled settings keep the powder from becoming too wet too quickly, though.
Safety Considerations and Contraindications
Wide ranges of doses of vitamin C are safe, with 2,000 mg per day for people being the highest amount that is considered acceptable. Liposomal transport doesn't change basic safety rules, but people with certain conditions may need different instructions. Because high doses of vitamin C make it easier for the body to absorb iron, people with hemochromatosis or other iron overload disorders should talk to their doctors before taking them. If someone has had kidney stones in the past, they should be closely watched. However, liposomal types of ascorbic acid cause less oxalate to form than regular ascorbic acid.
Procurement Strategies for Liposomal Vitamin C Powder
To get around in global supply chains, you need to be smart about choosing vendors, checking quality, and predicting market trends. Direct connections with manufacturers get rid of markups that distributors add on top of the price, and they also make the production process and quality control systems more open. Our offices in San Diego makes it easy for us to work with brands in the US by providing regional inventory, technical support, and legal advice that is tailored to the needs of the North American market.
Sourcing Channels and Negotiation Tactics
There are usually breaks at 100 kg, 500 kg, and 1,000 kg or more order amounts in wholesale price structures. Negotiating payment terms should strike a balance between managing cash flow and reducing supply risk. Letters of credit protect both parties in foreign transactions, and net-30 or net-60 terms award long-term relationships. Sample review programs let you try formulations before you commit to making a lot of them. This lowers the risk of developing new product ideas.
Quality Verification and Regulatory Compliance
According to ICH standards, a study of the Certificate of Analysis should show that the vitamin C content is at least 99%, the particle size distribution is within certain ranges (usually 50–200nm), the encapsulation efficiency is at least 85%, and there are no heavy metals, microbes, or solvents left over. Stability data showing that potency is maintained under rapid and real-time settings supports claims about shelf life and storage requirements.
Future Market Trends and Innovation Opportunities
The liposomal delivery market is still changing because of new tailored release mechanisms, combination treatments that work on multiple health paths, and efforts to find renewable sources of materials. More people are learning about bioavailability science, which leads to price premiumization, where brands set their own prices by showing they are more effective. Recent studies on vitamin C's part in immune memory, skin health, and metabolic function have opened up new uses for the vitamin beyond its standard cold and flu role.
Conclusion
Liposomal vitamin C powder fixes basic problems in nutrient delivery by encasing nutrients in phospholipids, which increases their absorption while also making them more stable and easier to handle. Product performance and market success are decided by procurement choices that balance technical standards, regulatory compliance, and supplier capabilities. EmerWell's EncapsWell™ platform offers cGMP-certified and scientifically proven formulations along with full OEM/ODM services, giving brands the confidence to launch unique goods. Businesses can take advantage of the rising demand for science-backed, high-efficacy health solutions by forming strategic sourcing relationships that focus on quality assurance, customization options, and market compliance.
FAQ
Is Long-Term Liposomal Vitamin C Use Safe?
Liposomal forms of vitamin C keep the safety profile that has been set for regular vitamin C over decades of study and practical use. If healthy people take between 500 and 2,000 mg every day, there isn't much risk, and liposomal delivery lowers the stomach problems that come with high doses of standard kinds. Continuous supplementation tests that last 12 months or more show that, when used correctly, it doesn't have any negative effects on kidney function, iron absorption, or other physiological factors.
How Much Better Is Liposomal Absorption Than Regular Tablets?
Comparative pharmacokinetics studies show that liposomal vitamin C has plasma amounts that are 8 times higher than similar doses of powder that is not encapsulated. The effects last all day because the blood levels stay high. This means that vitamin C is better able to enter cells, where it can do its job as an antioxidant, help make collagen, and regulate the immune system. This leads to clearly better results in clinical measures.
What Advantages Do Organic Formulations Offer?
Organic certification is mostly about rules for where to get raw materials, not about the molecular structure of vitamin C, which is the same in both synthetic and organically produced forms. People's preferences for organic ingredients affect how brands are seen and how much they can be charged, but bioavailability and effectiveness rely more on the quality of the liposomes that hold the ingredients than on whether they are organic or not.
Partner with EmerWell for Premium Liposomal Vitamin C Powder Supply
Through our own EncapsWell™ platform, EmerWell specializes in making high-bioavailability liposomal formulations. We can turn your product ideas into supplements that are ready to go to market and have been shown to deliver to cells. Our cGMP-certified production, full approvals such as HALAL, Kosher, and Non-GMO verification, and flexible OEM/ODM services help brands from formulating new products to making them available to more people. With our main office in San Diego and warehousing in the US, we offer fast sampling, technical advice, and legal guides that are specifically designed to meet the needs of the North American market. Get in touch with our team at info@emerwell-bio.com to talk about how we can make liposomal vitamin C powder that fits your product vision and comes with the quality guarantee and supply predictability that builds trust with customers.
References
Carr, A.C., & Maggini, S. (2017). Vitamin C and Immune Function. Nutrients, 9(11), 1211.
Davis, J.L., Paris, H.L., Beals, J.W., et al. (2016). Liposomal-encapsulated Ascorbic Acid: Influence on Vitamin C Bioavailability and Capacity to Protect Against Ischemia-Reperfusion Injury. Nutrition and Metabolic Insights, 9, 25-30.
Lykkesfeldt, J., & Tveden-Nyborg, P. (2019). The Pharmacokinetics of Vitamin C. Nutrients, 11(10), 2412.
Padayatty, S.J., Sun, H., Wang, Y., et al. (2004). Vitamin C Pharmacokinetics: Implications for Oral and Intravenous Use. Annals of Internal Medicine, 140(7), 533-537.
Hickey, S., Roberts, H.J., & Miller, N.J. (2008). Pharmacokinetics of Oral Vitamin C. Journal of Nutritional & Environmental Medicine, 17(3), 169-177.
Pires, L.R., Figueiredo, R.T., & Cabral, L.M. (2021). Liposomal Encapsulation as a Strategy to Improve the Bioavailability of Bioactive Compounds: A Review. Journal of Food Science and Technology, 58(11), 4139-4151.
Online Message
YOU MAY LIKE
High quality furniture manufacturer



_1776234307158.webp)





