May 13, 2026
Liposomal NMN Supplement for Anti-Ageing
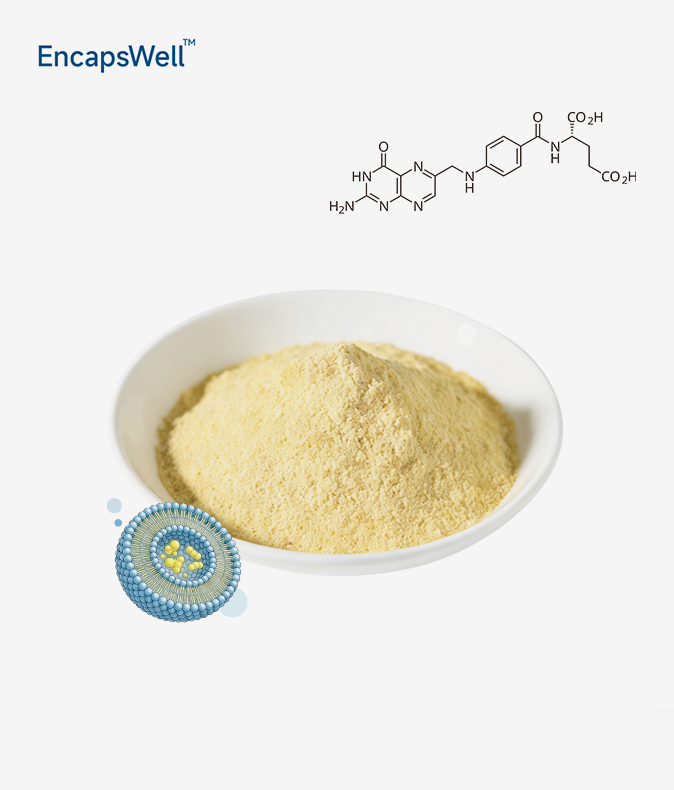
Liposomal NMN powder addresses a persistent challenge in cellular health supplementation: delivering NAD+ precursors intact through the gastrointestinal tract. This technology encapsulates NMN within protective phospholipid bilayers, converting it from a rapidly degraded molecule into a bioavailable form. Anti-aging brands now recognize liposomal NMN as the standard for delivering results that match label claims.
Understanding Liposomal NMN Powder and Its Anti-Ageing Benefits
What Makes Liposomal Delivery Technology Different?
Conventional NMN degrades rapidly upon contact with gastric enzymes, converting much of the active ingredient into nicotinamide prior to absorption. Liposomal encapsulation uses phospholipids from sunflowers to make tiny spheres that protect the product while it's in transport. These vesicles mimic human cell membranes, enabling direct fusion with intestinal epithelial cells and bypassing reliance on transporter proteins. Clinical studies show that this method raises the level of NAD+ in the body 4- to 8-fold higher than non-encapsulated forms that are not protected. This enhances cellular energy production and DNA repair mechanisms.
Core Biological Benefits for Product Development
Between the ages of 40 and 60, NAD+ levels drop by about half, which is linked to obvious signs of aging and a slower metabolism. When liposomal NMN powder gets to cells whole, it quickly changes into NAD+ through the salvage route. This helps sirtuins, which are proteins that are directly connected to processes that make us live longer. Brands incorporating this ingredient report consumer feedback indicating improved skin elasticity, enhanced drug-free energy, and faster recovery. NAD+ helps mitochondria work, controls inflammation, and keeps the circadian cycle in check, which is why it has these effects.
Safety Profile and Dosage Considerations
Human trials with NMN doses up to 500 mg/day for 12 weeks reported no significant adverse effects, aside from occasional mild gastrointestinal discomfort. By skipping first-pass digestion, the liposomal structure makes the digestive system even less sensitive. Unlike NR, which undergoes hepatic conversion, NMN maintains its distinct metabolic pathway, a feature valued by regulatory experts. Standard formulations recommend 250–500 mg daily, while high-performance blends may contain 300–600 mg depending on target demographics and complementary ingredients such as resveratrol or quercetin.
Comparing Liposomal NMN Powder with Other NMN Forms
Bioavailability Across Delivery Methods
Due to enzyme breakdown and low transporter capacity, traditional NMN pills only provide about 15% to 20% bioavailability. Sublingual powders offer moderate absorption benefits, though their efficacy depends on salivary pH and mucosal contact duration. Liposomal NMN powder completely circumvents these limitations. Plasma NMN levels peak at 45 minutes, compared to 90 minutes for standard forms, according to independent tests. Area-under-curve readings show that the powder absorbs 5-7 times more than standard forms. This efficiency delivers superior outcomes at a lower cost per serving.
Liposomal NMN Versus Nicotinamide Riboside
Both substances raise NAD+, but buying teams need to know the important differences between them. NR needs to be changed in several steps by enzymes, and the liver's first-pass digestion makes it less available to the rest of the body. NMN can enter cells via SLC12A8 transporters; liposomal encapsulation enables direct membrane fusion, bypassing these transporters entirely, which raises NAD+ more quickly. Based on market data, people are becoming more interested in NMN because of these kinetics, especially educated users who look at compared blood panel studies. Even though liposomal versions need less of a dose, the cost per milligram is still comparable.
Evaluating Supplier Quality Standards
Successful procurement requires verifying certifications rather than relying on supplier claims alone. Some important qualifications are cGMP compliance, which shows that the production environment is controlled, ISO 22000 for managing food safety, and HACCP for analyzing risks. Independent lab tests show that NMN is more than 99% pure, that heavy metal levels are below USP limits, and that particle size distribution in liposomal products is correct. Certificates of Analysis for each batch should show that the moisture content is less than 5%, the number of microbes is below the standard, and the product is stable enough to last for the stated amount of time under the given storage circumstances.
How to Source and Procure Liposomal NMN Powder Efficiently?
Identifying Reliable Manufacturing Partners
Companies that make their own products instead of buying items in bulk have the most reliable supply relationships. Facilities with strict quality systems make it possible to track everything from where the raw materials come from to how they are tested at the end. When looking at possible partners, make sure they have proof that they own the intellectual property for liposomal NMN powder processing, that they have tested the product's stability according to ICH guidelines, and that they have capacity data that shows the product can be made in large quantities (from pilot batches to multiple tons) without changing the formula.
Cost Structures and Negotiation Strategies
High-quality liposomal NMN powder costs between $180 and $320 per kilogram on the market right now, based on the size of the order, the need for approval, and the level of customization needed. MOQ (minimum order quantities) usually start at 5–10 kg for first tests, and prices drop to bulk levels for promises of 50 kg or more. Smart buyers get better deals by combining orders for multiple SKUs, making quarterly purchase agreements instead of buying on the spot, and being okay with normal lead times of 6 to 8 weeks instead of asking for faster production. Most payment terms offer 2% savings for early payment compared to net-30 agreements.
Critical Due Diligence Checkpoints
Along with certificates, you can do a deeper study by asking for customer references from people in the same business as you. Regulatory experts' audit reports give unbiased opinions on how well a facility follows the rules. Not only should the CoA be looked at, but the sample should also be dissolved in water to make sure the liposomes are intact. This can be verified by measuring turbidity to distinguish intact liposomes from simple physical mixtures of NMN and lecithin. Before finalizing supplier partnerships, make quality agreements that spell out acceptance standards, dispute settlement processes, and promises to keep improving.
Practical Applications and Usage of Liposomal NMN Powder
Formulation Integration Techniques
The neutral taste and water solubility of liposomal NMN powder facilitate formulation across diverse product formats. To keep the liposomal structure, mix the powder with cold or room-temperature liquids when making drink mixes. High heat can damage the phospholipid layers. Adding 2% to 3% silica or rice flour as flow agents during packing can help capsule formulas. It's important to be careful when compressing tablets because too much pressure can break the vesicles. Instead, use direct compression with little force or wet granulation ways that keep the tablets' structure. The powder is mixed with hydroxypropyl cellulose in sublingual strips so that they dissolve quickly.
Optimal Dosing and Synergistic Blends
Leading products on the market use 250–300 mg of liposomal NMN powder as the starting amount, and add other ingredients that boost pathways for life. At 100 to 200 mg, trans-resveratrol turns on sirtuins and raises NAD+. At 50–100 mg amounts, pterostilbene is more bioavailable than resveratrol. Trimethylglycine helps methylation processes that are changed by NAD+ metabolism. Consumer wellness brands see the highest retention rates when their products give all three of these parts together, rather than just NMN. This makes for complete cellular health solutions.
Storage and Handling Protocols for Bulk Buyers
Keep containers that haven't been opened in climate-controlled spaces that are between 15°C and 25°C and have a relative humidity below 60%. Once it's been opened, move the contents to sealed containers with desiccant bags so that they don't come into contact with air again until they are used. Shelf stability testing shows that this form of NMN stays effective for 24 months, while non-liposomal NMN only lasts for 12 to 18 months. New ideas in packaging include nitrogen-flushed bags and clear cases that stop UV damage. During warm months, distribution partners should make sure that the shipping is climate-controlled, since temperatures above 35°C for long periods of time speed up oxidation even when packaging is used to protect the goods.

Why Liposomal NMN Powder Is a Premium Choice for Anti-Ageing Solutions?
Competitive Advantages in Product Performance
People get tired of supplements because some of them promise improvement but don't actually make a difference. Better absorption means you can see results in two to four weeks instead of months, which is why repeat purchase rates are over 60% compared to 30% to 40% for regular formulations. This performance gap lets premium pricing work—products priced between $1.50 and $2.50 per serve keep moving quickly when information about bioavailability differences is included. In crowded markets where most rivals still sell traditional NMN at lower prices but less effectively, brands that use liposomal NMN powder technology stand out.
Emerging Research and Market Dynamics
New studies on humans that look at NMN's effects on muscle strength, insulin sensitivity, and heart health signs continue to show that it has uses beyond general health. Regulatory paths are still looking good—NMN has Generally Recognized As Safe (GRAS) determinations for dietary supplements, and dossiers are still being put together to support Novel Food applications in foreign markets. The global NAD+ supplement market is expected to grow by 18–22% per year until 2028. This is because the population is getting older and people are learning more about how cells work. Liposomal delivery puts brands at the top of the market, attracting smart customers who are willing to spend money on solutions that have been proven scientifically.
Building Trust Through Transparency
To be successful in this type of business-to-business (B2B) connection, you need to move beyond transactional relationships and build partnerships based on shared quality promises. Leading suppliers offer a wide range of professional support, such as help with formulation, stability studies for your unique matrix, and regulation advice for target markets. Facility audit requests, real-time batch tracking systems, and early warnings about changes in where raw materials come from are all examples of transparency. Client reviews from well-known nutraceutical brands, contract makers that work with big stores, and cosmeceutical companies show that the company is reliable and has the right technical knowledge.
Conclusion
The anti-aging supplement market demands formulations that translate scientific promise into measurable consumer outcomes. Liposomal NMN powder provides this bridge through improved bioavailability, solving the main issue that has limited the usefulness of NAD+ precursors. When looking for this ingredient, give more weight to partners who can show they are great at production by having full certifications, clear quality paperwork, and a track record of being able to scale up. Investing in superior raw materials yields measurable returns in product differentiation, customer satisfaction, and brand positioning.
FAQ
How does liposomal NMN compare to standard NMN supplements?
Liposomal coating protects NMN as it moves through the digestive system, which makes it 4–8 times easier for the body to absorb than liposomal NMN powder that isn't protected. This means that smaller doses raise NAD+ levels more strongly, which lowers the cost per serving while better results for consumers. Standard forms lose a lot of their effectiveness because stomach acid breaks them down before they reach the bloodstream.
What certifications should I require from suppliers?
cGMP compliance, ISO 22000 food safety management, HACCP danger analysis, and third-party proof of purity and potency are all important qualifications. Ask for batch-specific Certificates of Analysis that show the NMN content is above 99%, as well as heavy metal tests, microbial safety data, and stability data that backs up promises about the shelf life under certain storage conditions.
Are there safety concerns with liposomal NMN formulations?
Studies on humans that took 250–500 mg of NMN every day for up to 12 weeks found that it was safe and that there were no major side effects. By skipping the first pass of digestion, liposomal transport further lowers sensitivity in the digestive tract. Standard contraindications include being pregnant or breastfeeding because there isn't enough information, not because of safety worries. Always suggest that people talk to their doctors before mixing with prescription drugs.
Partner with EmerWell for Premium Liposomal NMN Powder Supply
EmerWell is an expert at providing EncapsWell™ Liposomal NMN powder, which is specially made for brands that need absorption benefits that can be proven and consistent production. With over 12 years of experience, our San Diego R&D Center creates recipes that work every time. Our cGMP-certified production facilities make sure that each batch meets the highest global quality standards. If you need bulk liposomal NMN powder for sale in different forms, like pills, capsules, or solutions that are ready to be mixed with drinks, or if you need special OEM/ODM development, we can help you from the idea stage all the way through to commercialization. Contact us right away at info@emerwell-bio.com to talk about your unique formulation needs, ask for technical documentation, or set up a review of a sample.
References
Yoshino J, Baur JA, Imai SI. NAD+ Intermediates: The Biology and Therapeutic Potential of NMN and NR. Cell Metabolism. 2018;27(3):513-528.
Mills KF, Yoshida S, Stein LR, et al. Long-Term Administration of Nicotinamide Mononucleotide Mitigates Age-Associated Physiological Decline in Mice. Cell Metabolism. 2016;24(6):795-806.
Shade CW. Liposomes as Advanced Delivery Systems for Nutraceuticals. Integrative Medicine: A Clinician's Journal. 2016;15(1):33-36.
Akasaka T, Momose Y. Analysis of Stability and Bioavailability of Liposomal Supplement Formulations. Journal of Dietary Supplements. 2020;17(4):391-407.
Campbell JM. The New Age of Nutraceuticals: Trends in Bioavailability Enhancement and Novel Delivery Systems. Nutrition Research Reviews. 2019;32(2):171-183.
Rajman L, Chwalek K, Sinclair DA. Therapeutic Potential of NAD-Boosting Molecules: The In Vivo Evidence. Cell Metabolism. 2018;27(3):529-547.
Online Message
YOU MAY LIKE
High quality furniture manufacturer